West of Scotland Paediatric Hepatitis Pathway
exp date isn't null, but text field is
Objectives
Public Health Scotland (PHS) and the UK Health Security Agency (UKHSA)are currently investigating an outbreak of idiopathic hepatitis in children.1,2 There are many similarities to viral hepatitis, but no single agent has been identified as causative. This guideline summarises case definitions, referral pathway, and initial recommended investigations for children presenting with idiopathic hepatitis in this public health context. It also outlines reporting responsibilities, and the process of recruitment to the ISARIC study as recommended by PHS.
Scope
This is a consensus guidance document for paediatric units whose tertiary referral centre is the Royal Hospital for Children, Glasgow and is approved by the West of Scotland Gastroenterology, Hepatology and Nutrition Managed Clinical network . It is informed by and designed to complement guidance produced by Public Health Scotland. It is not intended as guidance for primary care. Children with jaundice or who have been identified as having transaminitis in primary care settings should be discussed urgently with their local paediatric unit.
Confirmed
A person presenting with a serum transaminase greater than 500 iu/l (AST or ALT) without any known cause*, who is 10 years of age and under or a contact of any age of a possible or confirmed case, since 1 January 2022.
*If hepatitis A-E serology, CMV and EBV results are awaited, but other criteria are met, the case should be reported to PHS but will be classified as “pending classification”. Cases that meet the initial criteria but are later found to have alternative causes will be denotified.
This is a working case definition and subject to change. Note the PHS definition differs from the UKHSA definition. Please refer to the latest PHS updates to confirm. There should also be a low threshold for discussing with the GI team at RHC Glasgow any child up to 16 years who presents with transaminases >500 IU/L with no alternative diagnosis.
The ISARIC study case definition is wider and children up to 16 years with ALT or AST >400IU/L can be recruited to the study. Children who meet this definition, but not the PHS definition should not be reported as cases to public health.
The referral pathway is summarised in Figure 1. Paediatric Units who identify potential cases should complete the referral proforma (Appendix 1 or link here) and contact the GI Consultant on call at RHC Glasgow for an initial case discussion. After this discussion, confirmed cases should be reported by the referring paediatric unit to their local health protection team (HPT). Please see Appendix 2 for list of HPT contacts by locality/postcode. The most appropriate initial place/team of care will be agreed. Recruitment to the ISARIC study will be undertaken by the local paediatric team if remaining in general paediatric care, or by the GI team at RHC if transferred. Initial and long-term follow-up should be agreed in discussion with the GI team (lead clinician for hepatology Dr Rachel Tayler).
DRAFT West of Scotland Paediatric Hepatitis Pathway Version 1.1 10th May 2022
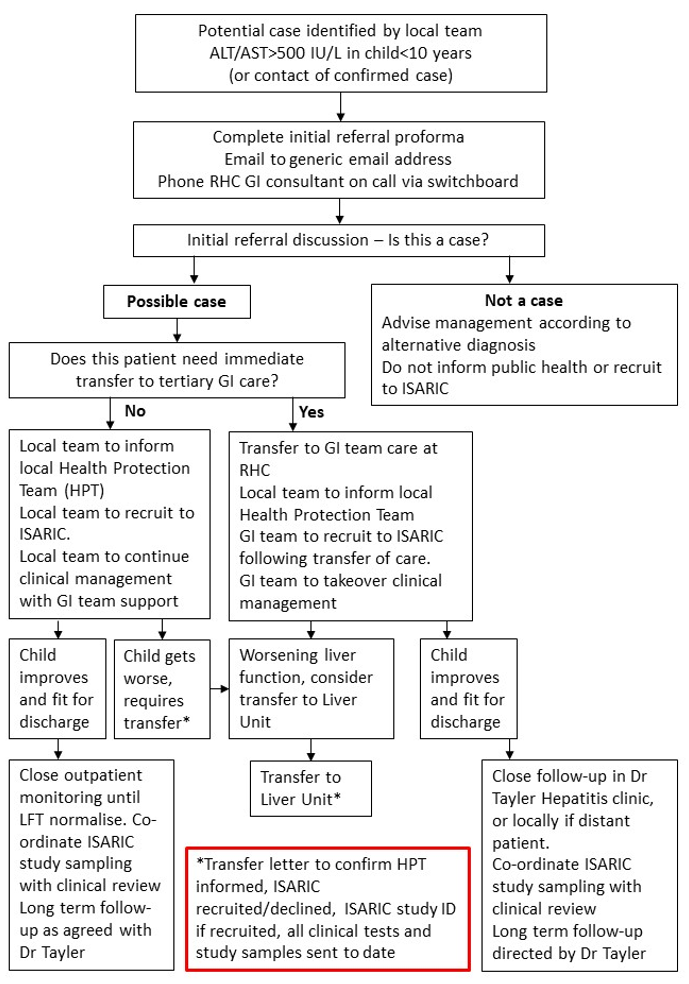
Figure 1: West of Scotland Referral Pathway for Paediatric Hepatitis
Investigations recommended by Public Health Scotland are listed in Tables 1 and 2. Where sampling is difficult, please prioritise blood tests essential to clinical monitoring, viral and autoimmune hepatitis diagnostics and stool and respiratory samples for virology.
Informing relevant laboratories
It is essential that biochemistry, virology and immunology laboratories are aware that these are samples from patients who are being investigated as part of this public health response. Samples are being processed and excess stored in specific ways for this investigation. Please include “Query non A-E Hepatitis Investigation” in the clinical details of all samples.
Please phone the duty biochemist at your hospital to enable them to store any excess sample appropriately for future investigation.
Please inform virology and immunology laboratories by email of the name and CHI number of each patient and confirm investigation for Non A-E hepatitis ( email west-ssvc2@ggc.scot.nhs.uk and immunology.labs@ggc.scot.nhs.uk).
Table 1: Initial Investigations - All patients
|
Initial tests (all patients) |
||
|
Sample Type |
Test |
Sent |
|
Virology |
|
|
|
Blood PCR (1x 5ml EDTA) |
CMV, EBV, adenovirus PCR, HSV PCR, enterovirus/parechovirus PCR |
|
|
BBV/Serology (1x 5ml EDTA or 3x 1.5ml EDTA) |
Hepatitis A IgM, Hepatitis B sAg, Hepatitis E IgM, HCV antibody, HCV PCR, CMV IgM, EBV IgM, Sars-Cov-2 serology |
|
|
Throat swab PCR |
Standard and “immunocompromised respiratory virus PCR panel (including adenovirus/enterovirus/influenza, SARS-CoV2) |
|
|
Stool PCR |
Adenovirus, sapovirus, norovirus, enterovirus (request faecal virus PCR and enterovirus PCR) |
|
|
Microbiology |
|
|
|
Stool |
Bacterial culture |
|
|
Haematology |
|
|
|
EDTA |
FBC incl Retics |
|
|
Lithium Heparin / Serum |
Blood film, DCT |
|
|
Coagulation Tube |
Coag |
|
|
Biochemistry |
|
|
|
Lithium Heparin / Serum Min vol 1ml |
UE +CRP Bone profile and Magnesium LFT including AST GGT Amylase Cholesterol/trigs Urate Ammonia Paracetamol level |
|
|
Fluoride Oxalate Min vol 0.2ml |
Glucose, Lactate |
|
|
Urine (plain universal container) 5ml |
Urine electrolytes |
|
|
Lithium Heparin / Serum Min vol 2ml |
Alpha-1-antitrypsin phenotype |
|
|
Lithium Heparin / Serum Min vol 1ml |
Copper & caeruloplasmin |
|
|
Lithium Heparin / Serum Min vol 0.4ml |
Alpha fetoprotein |
|
|
Serum (Lithium heparin) Min vol 0.4ml |
Ferritin |
|
|
Lithium Heparin / Serum Min vol 1ml |
CK, TFTs, LDH, cortisol |
|
|
EDTA (1ml) |
HBA1C |
|
|
Immunology |
|
|
|
Lithium Heparin / Serum Min vol 0.4ml |
Immunoglobulins (for IgG level) |
|
|
Serum |
“Liver abs” autoantibody screen (mitochondrial, smooth muscle, LKM) |
|
|
Serum |
ANA (anti-nuclear and centromere antibodies) |
|
|
Serum |
Coeliac screen |
|
|
Imaging |
|
|
|
Ultrasound |
Baseline liver US |
|
Table 2: Additional tests if clinically indicated
|
Additional tests only if clinically indicated |
|
|
Features of streptococcal infection (typical rash, sore throat) |
Throat swab for c/s, ASOT |
|
Fever/suspected sepsis |
Blood cultures |
|
Suspected metabolic condition (discuss with metabolic team) |
Plasma amino and urine organic acids |
|
Suspected vitamin deficiency |
Vitamin A, D and E |
|
Suspected leptospirosis (history of freshwater swimming, contact rats/mice or farm animals, clinical features include conjunctivitis, headache and cough) |
Leptospirosis PCR (blood and urine) and serology (blood) |
|
Toxicology (only after discussion with National Poisons Information Service) |
Lead, mercury, iron (not ferritin), arsenic, mercury, urinary amatoxin for amanita (mushroom), longer exposure tests for paracetamol |
Cases should remain under general paediatric care unless concerns regarding liver synthetic function or complications. Patients should be discussed regularly with on call GI consultant.
If the patient is jaundiced or has a raised bilirubin on bloods, please commence oral ursodeoxycholic acid (10mg/kg twice daily, increased if necessary to three times daily) and oral vitamin K (0.3mg/kg, max 10mg). Cases with prolonged PT >15 should be discussed with GI to consider IV vitamin K and other supportive management.
The on call gastroenterology consultant available ~8-5 Monday to Friday on 85806 or 24 hours a day via switchboard.
LFT, GGT, U&E, Bone profile, magnesium and coagulation studies should be checked daily initially. Blood glucose monitoring should be at least daily, and more frequent if concerns. The most important marker of synthetic liver function is PT. FBC should be monitored at least twice weekly.
Daily venous bloods will be required until bilirubin, PT, ALT and AST are all falling and the child is no longer requiring intravenous vitamin K. Step down from daily bloods to less frequent monitoring, and discharge decisions should be discussed with the GI consultant on call.
Following discharge, patients should be followed up by Dr Tayler in the dedicated hepatitis clinic, or locally as agreed with Dr Tayler.
ISARIC-CCP-UK Study
PHS is recommending recruitment of all identified cases to the ISARIC-CCP-UK study. There are 3 “tiers” to the ISARIC study. Tier 0 collects data and allows for use of excess clinical samples, Tier 1 collects research samples only at recruitment and Tier 2 collects serial samples on days 3, 9 and 28. For this outbreak, the most important research samples are the stool and viral throat swabs which should be obtained as soon as possible after consent. Use of excess clinical samples is also a priority, so even if families decline “Tier 1 or 2” sampling, Tier 0 consent should be encouraged. All other blood tests should be timed to co-incide with clinical sampling where possible to minimise the number of times children are bled. A detailed guide to ISARIC samples is available here. Details of the ISARIC study and all study documents can be found here: https://isaric4c.net/protocols/. See Appendix 3 for a visual guide to samples required for ISARIC. Blood sampling should be co-ordinated with clinical sampling where possible.
Please inform the local ISARIC study team by email as soon as possible when cases are identified (see Appendix 3).
|
Appendix 1. Contact details for NHS Board Health Protection Teams NHS Board |
Office Hours Telephone Number |
Out of Hours Telephone Number |
Generic email address |
|
Ayrshire and Arran |
01563 521133 and ask for Public Health On Call |
||
|
Borders |
|||
|
Dumfries and Galloway |
|||
|
Fife |
|||
|
Forth Valley |
|||
|
Grampian |
|||
|
Greater Glasgow & Clyde |
0141 201 4917 public 4927 HPS |
||
|
Highland |
|||
|
Lanarkshire |
|||
|
Lothian |
0131 242 1000 and ask for public health on call |
||
|
Orkney |
|||
|
Shetland |
|||
|
Tayside |
|||
|
Western Isles |
01851 704704 (ask for on-call public health) |
|
Site |
PI |
|
Lead research nurse |
|
|
Ayrshire& Arran |
Lawrence Armstrong |
Lawrence.Armstrong@aapct.scot.nhs.uk |
Claire Bell |
Claire.Bell@aapct.scot.nhs.uk |
|
Borders |
Clare Irving |
Clare.Irving@borders.scot.nhs.uk |
Joy Dawson |
joy.dawson@borders.scot.nhs.uk |
|
Dumfries & Galloway |
Andrew Mitra |
andrew.mitra@nhs.scot |
Janie Candlish |
janie.candlish@nhs.scot |
|
Fife |
John Morrice & Helen Brotherton |
John.morrice@nhs.scot Helen.brotherton@nhs.scot |
Laura Beveridge |
Laura.Beveridge2@nhs.scot |
|
Forth Valley |
Kristyna Bohmova Sheena Logan |
kristyna.bohmova@nhs.scot sheena.logan@nhs.scot |
Laura McGenily |
laura.mcgenily@nhs.scot |
|
GGC |
Louisa Pollock |
louisa.pollock@ggc.scot.nhs.uk |
Barry Milligan Naomi Hickey |
Barry.milligan@ggc.scot.nhs.uk Naomi.Hickey@ggc.scot.nhs.uk |
|
Grampian |
Shyla Kishore |
shyla.kishore@nhs.scot |
Maggie Connon Catriona Ward |
Margaret.connon@nhs.scot catriona.ward@nhs.scot |
|
Highland |
David Goudie |
David.goudie2@nhs.scot |
tbc |
|
|
Lanarkshire |
Antonia Ho |
Antonia.ho@lanarkshire.scot.nhs.uk |
Suzanne Clements |
Suzanne.clements@lanarkshire.scot.nhs.uk |
|
Lothian |
Paul Henderson |
Paul.henderson@nhslothian.scot.nhs.uk |
Maxine Ramsay |
Maxine.ramsay@nhslothian@scot.nhs.uk |
|
Tayside |
Louise Gannon (tbc) |
Louise.gannon@nhs.scot |
Susan Macfarlane |
Susan.macfarlane3@nhs.scot |
- Marsh Kimberly, Tayler Rachel, Pollock Louisa, Roy Kirsty, Lakha Fatim, Ho Antonia, Henderson David, Divala Titus, Currie Sandra, Yirrell David, Lockhart Michael, Rossi Maria K., Phin Nick. Investigation into cases of hepatitis of unknown aetiology among young children, Scotland, 1 January 2022 to 12 April 2022. Euro Surveill. 2022;27(15):pii=2200318.
- UK Health Security Agency. Increase in hepatitis (liver inflammation) cases in children under investigation: Regular UKHSA updates on the ongoing investigation into higher than usual rates of liver inflammation (hepatitis) in children across the UK Last accessed 10/05/2022
Last reviewed: 10 May 2022
Next review: 11 May 2025
Author(s): Rachel Tayler Louisa Pollock Richard Hansen
Version: 1

